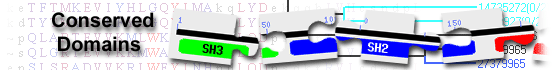rho GTPase-activating protein 27 isoform e [Homo sapiens]
List of domain hits

| Name | Accession | Description | Interval | E-value | ||||
| RhoGAP_ARHGAP27_15_12_9 | cd04403 | RhoGAP_ARHGAP27_15_12_9: GTPase-activator protein (GAP) domain for Rho-like GTPases found in ... |
673-859 | 1.94e-128 | ||||
RhoGAP_ARHGAP27_15_12_9: GTPase-activator protein (GAP) domain for Rho-like GTPases found in ARHGAP27 (also called CAMGAP1), ARHGAP15, 12 and 9-like proteins; This subgroup of ARHGAPs are multidomain proteins that contain RhoGAP, PH, SH3 and WW domains. Most members that are studied show GAP activity towards Rac1, some additionally show activity towards Cdc42. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. : Pssm-ID: 239868 [Multi-domain] Cd Length: 187 Bit Score: 382.89 E-value: 1.94e-128
|
||||||||
| PH_ARHGAP9-like | cd13233 | Beta-spectrin pleckstrin homology (PH) domain; ARHGAP family genes encode Rho/Rac/Cdc42-like ... |
477-591 | 3.52e-46 | ||||
Beta-spectrin pleckstrin homology (PH) domain; ARHGAP family genes encode Rho/Rac/Cdc42-like GTPase activating proteins with RhoGAP domain. The ARHGAP members here all have a PH domain upstream of their C-terminal RhoGAP domain. Some have additional N-terminal SH3 and WW domains. The members here include: ARHGAP9, ARHGAP12, ARHGAP15, and ARHGAP27. ARHGAP27 and ARHGAP12 shared the common-domain structure, consisting of SH3, WW, PH, and RhoGAP domains. The PH domain of ArhGAP9 employs a non-canonical phosphoinositide binding mechanism, a variation of the spectrin- Ins(4,5)P2-binding mode, that gives rise to a unique PI binding profile, namely a preference for both PI(4,5)P2 and the PI 3-kinase products PI(3,4,5)P3 and PI(3,4)P2. This lipid binding mechanism is also employed by the PH domain of Tiam1 and Slm1. PH domains have diverse functions, but in general are involved in targeting proteins to the appropriate cellular location or in the interaction with a binding partner. They share little sequence conservation, but all have a common fold, which is electrostatically polarized. Less than 10% of PH domains bind phosphoinositide phosphates (PIPs) with high affinity and specificity. PH domains are distinguished from other PIP-binding domains by their specific high-affinity binding to PIPs with two vicinal phosphate groups: PtdIns(3,4)P2, PtdIns(4,5)P2 or PtdIns(3,4,5)P3 which results in targeting some PH domain proteins to the plasma membrane. A few display strong specificity in lipid binding. Any specificity is usually determined by loop regions or insertions in the N-terminus of the domain, which are not conserved across all PH domains. PH domains are found in cellular signaling proteins such as serine/threonine kinase, tyrosine kinases, regulators of G-proteins, endocytotic GTPases, adaptors, as well as cytoskeletal associated molecules and in lipid associated enzymes. : Pssm-ID: 270053 Cd Length: 110 Bit Score: 160.52 E-value: 3.52e-46
|
||||||||
| SH3_ARHGAP27 | cd12069 | Src Homology 3 domain of Rho GTPase-activating protein 27; Rho GTPase-activating proteins ... |
10-66 | 3.54e-33 | ||||
Src Homology 3 domain of Rho GTPase-activating protein 27; Rho GTPase-activating proteins (RhoGAPs or ARHGAPs) bind to Rho proteins and enhance the hydrolysis rates of bound GTP. ARHGAP27, also called CAMGAP1, shows GAP activity towards Rac1 and Cdc42. It binds the adaptor protein CIN85 and may play a role in clathrin-mediated endocytosis. It contains SH3, WW, Pleckstin homology (PH), and RhoGAP domains. SH3 domains bind to proline-rich ligands with moderate affinity and selectivity, preferentially to PxxP motifs; they play a role in the regulation of enzymes by intramolecular interactions, changing the subcellular localization of signal pathway components and mediate multiprotein complex assemblies. : Pssm-ID: 213002 Cd Length: 57 Bit Score: 121.85 E-value: 3.54e-33
|
||||||||
| WW | pfam00397 | WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds ... |
247-278 | 1.71e-07 | ||||
WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds proline-rich peptide motifs in vitro. : Pssm-ID: 459800 [Multi-domain] Cd Length: 30 Bit Score: 47.88 E-value: 1.71e-07
|
||||||||
| PRP40 super family | cl34905 | Splicing factor [RNA processing and modification]; |
239-376 | 5.16e-05 | ||||
Splicing factor [RNA processing and modification]; The actual alignment was detected with superfamily member COG5104: Pssm-ID: 227435 [Multi-domain] Cd Length: 590 Bit Score: 47.00 E-value: 5.16e-05
|
||||||||
| WW | pfam00397 | WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds ... |
415-442 | 1.71e-03 | ||||
WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds proline-rich peptide motifs in vitro. : Pssm-ID: 459800 [Multi-domain] Cd Length: 30 Bit Score: 36.71 E-value: 1.71e-03
|
||||||||
| Name | Accession | Description | Interval | E-value | ||||
| RhoGAP_ARHGAP27_15_12_9 | cd04403 | RhoGAP_ARHGAP27_15_12_9: GTPase-activator protein (GAP) domain for Rho-like GTPases found in ... |
673-859 | 1.94e-128 | ||||
RhoGAP_ARHGAP27_15_12_9: GTPase-activator protein (GAP) domain for Rho-like GTPases found in ARHGAP27 (also called CAMGAP1), ARHGAP15, 12 and 9-like proteins; This subgroup of ARHGAPs are multidomain proteins that contain RhoGAP, PH, SH3 and WW domains. Most members that are studied show GAP activity towards Rac1, some additionally show activity towards Cdc42. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239868 [Multi-domain] Cd Length: 187 Bit Score: 382.89 E-value: 1.94e-128
|
||||||||
| RhoGAP | smart00324 | GTPase-activator protein for Rho-like GTPases; GTPase activator proteins towards Rho/Rac ... |
686-860 | 8.84e-64 | ||||
GTPase-activator protein for Rho-like GTPases; GTPase activator proteins towards Rho/Rac/Cdc42-like small GTPases. etter domain limits and outliers. Pssm-ID: 214618 Cd Length: 174 Bit Score: 211.74 E-value: 8.84e-64
|
||||||||
| RhoGAP | pfam00620 | RhoGAP domain; GTPase activator proteins towards Rho/Rac/Cdc42-like small GTPases. |
689-835 | 8.35e-59 | ||||
RhoGAP domain; GTPase activator proteins towards Rho/Rac/Cdc42-like small GTPases. Pssm-ID: 459875 Cd Length: 148 Bit Score: 197.00 E-value: 8.35e-59
|
||||||||
| PH_ARHGAP9-like | cd13233 | Beta-spectrin pleckstrin homology (PH) domain; ARHGAP family genes encode Rho/Rac/Cdc42-like ... |
477-591 | 3.52e-46 | ||||
Beta-spectrin pleckstrin homology (PH) domain; ARHGAP family genes encode Rho/Rac/Cdc42-like GTPase activating proteins with RhoGAP domain. The ARHGAP members here all have a PH domain upstream of their C-terminal RhoGAP domain. Some have additional N-terminal SH3 and WW domains. The members here include: ARHGAP9, ARHGAP12, ARHGAP15, and ARHGAP27. ARHGAP27 and ARHGAP12 shared the common-domain structure, consisting of SH3, WW, PH, and RhoGAP domains. The PH domain of ArhGAP9 employs a non-canonical phosphoinositide binding mechanism, a variation of the spectrin- Ins(4,5)P2-binding mode, that gives rise to a unique PI binding profile, namely a preference for both PI(4,5)P2 and the PI 3-kinase products PI(3,4,5)P3 and PI(3,4)P2. This lipid binding mechanism is also employed by the PH domain of Tiam1 and Slm1. PH domains have diverse functions, but in general are involved in targeting proteins to the appropriate cellular location or in the interaction with a binding partner. They share little sequence conservation, but all have a common fold, which is electrostatically polarized. Less than 10% of PH domains bind phosphoinositide phosphates (PIPs) with high affinity and specificity. PH domains are distinguished from other PIP-binding domains by their specific high-affinity binding to PIPs with two vicinal phosphate groups: PtdIns(3,4)P2, PtdIns(4,5)P2 or PtdIns(3,4,5)P3 which results in targeting some PH domain proteins to the plasma membrane. A few display strong specificity in lipid binding. Any specificity is usually determined by loop regions or insertions in the N-terminus of the domain, which are not conserved across all PH domains. PH domains are found in cellular signaling proteins such as serine/threonine kinase, tyrosine kinases, regulators of G-proteins, endocytotic GTPases, adaptors, as well as cytoskeletal associated molecules and in lipid associated enzymes. Pssm-ID: 270053 Cd Length: 110 Bit Score: 160.52 E-value: 3.52e-46
|
||||||||
| SH3_ARHGAP27 | cd12069 | Src Homology 3 domain of Rho GTPase-activating protein 27; Rho GTPase-activating proteins ... |
10-66 | 3.54e-33 | ||||
Src Homology 3 domain of Rho GTPase-activating protein 27; Rho GTPase-activating proteins (RhoGAPs or ARHGAPs) bind to Rho proteins and enhance the hydrolysis rates of bound GTP. ARHGAP27, also called CAMGAP1, shows GAP activity towards Rac1 and Cdc42. It binds the adaptor protein CIN85 and may play a role in clathrin-mediated endocytosis. It contains SH3, WW, Pleckstin homology (PH), and RhoGAP domains. SH3 domains bind to proline-rich ligands with moderate affinity and selectivity, preferentially to PxxP motifs; they play a role in the regulation of enzymes by intramolecular interactions, changing the subcellular localization of signal pathway components and mediate multiprotein complex assemblies. Pssm-ID: 213002 Cd Length: 57 Bit Score: 121.85 E-value: 3.54e-33
|
||||||||
| PH | smart00233 | Pleckstrin homology domain; Domain commonly found in eukaryotic signalling proteins. The ... |
475-590 | 2.52e-09 | ||||
Pleckstrin homology domain; Domain commonly found in eukaryotic signalling proteins. The domain family possesses multiple functions including the abilities to bind inositol phosphates, and various proteins. PH domains have been found to possess inserted domains (such as in PLC gamma, syntrophins) and to be inserted within other domains. Mutations in Brutons tyrosine kinase (Btk) within its PH domain cause X-linked agammaglobulinaemia (XLA) in patients. Point mutations cluster into the positively charged end of the molecule around the predicted binding site for phosphatidylinositol lipids. Pssm-ID: 214574 [Multi-domain] Cd Length: 102 Bit Score: 55.25 E-value: 2.52e-09
|
||||||||
| WW | pfam00397 | WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds ... |
247-278 | 1.71e-07 | ||||
WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds proline-rich peptide motifs in vitro. Pssm-ID: 459800 [Multi-domain] Cd Length: 30 Bit Score: 47.88 E-value: 1.71e-07
|
||||||||
| WW | smart00456 | Domain with 2 conserved Trp (W) residues; Also known as the WWP or rsp5 domain. Binds ... |
246-278 | 2.40e-07 | ||||
Domain with 2 conserved Trp (W) residues; Also known as the WWP or rsp5 domain. Binds proline-rich polypeptides. Pssm-ID: 197736 [Multi-domain] Cd Length: 33 Bit Score: 47.60 E-value: 2.40e-07
|
||||||||
| WW | cd00201 | Two conserved tryptophans domain; also known as the WWP or rsp5 domain; around 40 amino acids; ... |
250-278 | 1.98e-06 | ||||
Two conserved tryptophans domain; also known as the WWP or rsp5 domain; around 40 amino acids; functions as an interaction module in a diverse set of signalling proteins; binds specific proline-rich sequences but at low affinities compared to other peptide recognition proteins such as antibodies and receptors; WW domains have a single groove formed by a conserved Trp and Tyr which recognizes a pair of residues of the sequence X-Pro; variable loops and neighboring domains confer specificity in this domain; there are five distinct groups based on binding: 1) PPXY motifs 2) the PPLP motif; 3) PGM motifs; 4) PSP or PTP motifs; 5) PR motifs. Pssm-ID: 238122 [Multi-domain] Cd Length: 31 Bit Score: 44.83 E-value: 1.98e-06
|
||||||||
| SH3_9 | pfam14604 | Variant SH3 domain; |
13-65 | 8.16e-06 | ||||
Variant SH3 domain; Pssm-ID: 434066 [Multi-domain] Cd Length: 49 Bit Score: 43.76 E-value: 8.16e-06
|
||||||||
| PH | pfam00169 | PH domain; PH stands for pleckstrin homology. |
477-589 | 2.75e-05 | ||||
PH domain; PH stands for pleckstrin homology. Pssm-ID: 459697 [Multi-domain] Cd Length: 105 Bit Score: 43.71 E-value: 2.75e-05
|
||||||||
| PRP40 | COG5104 | Splicing factor [RNA processing and modification]; |
239-376 | 5.16e-05 | ||||
Splicing factor [RNA processing and modification]; Pssm-ID: 227435 [Multi-domain] Cd Length: 590 Bit Score: 47.00 E-value: 5.16e-05
|
||||||||
| WW | pfam00397 | WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds ... |
415-442 | 1.71e-03 | ||||
WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds proline-rich peptide motifs in vitro. Pssm-ID: 459800 [Multi-domain] Cd Length: 30 Bit Score: 36.71 E-value: 1.71e-03
|
||||||||
| WW | smart00456 | Domain with 2 conserved Trp (W) residues; Also known as the WWP or rsp5 domain. Binds ... |
417-443 | 2.69e-03 | ||||
Domain with 2 conserved Trp (W) residues; Also known as the WWP or rsp5 domain. Binds proline-rich polypeptides. Pssm-ID: 197736 [Multi-domain] Cd Length: 33 Bit Score: 36.04 E-value: 2.69e-03
|
||||||||
| WW | pfam00397 | WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds ... |
301-328 | 7.06e-03 | ||||
WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds proline-rich peptide motifs in vitro. Pssm-ID: 459800 [Multi-domain] Cd Length: 30 Bit Score: 34.79 E-value: 7.06e-03
|
||||||||
| Name | Accession | Description | Interval | E-value | ||||
| RhoGAP_ARHGAP27_15_12_9 | cd04403 | RhoGAP_ARHGAP27_15_12_9: GTPase-activator protein (GAP) domain for Rho-like GTPases found in ... |
673-859 | 1.94e-128 | ||||
RhoGAP_ARHGAP27_15_12_9: GTPase-activator protein (GAP) domain for Rho-like GTPases found in ARHGAP27 (also called CAMGAP1), ARHGAP15, 12 and 9-like proteins; This subgroup of ARHGAPs are multidomain proteins that contain RhoGAP, PH, SH3 and WW domains. Most members that are studied show GAP activity towards Rac1, some additionally show activity towards Cdc42. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239868 [Multi-domain] Cd Length: 187 Bit Score: 382.89 E-value: 1.94e-128
|
||||||||
| RhoGAP | smart00324 | GTPase-activator protein for Rho-like GTPases; GTPase activator proteins towards Rho/Rac ... |
686-860 | 8.84e-64 | ||||
GTPase-activator protein for Rho-like GTPases; GTPase activator proteins towards Rho/Rac/Cdc42-like small GTPases. etter domain limits and outliers. Pssm-ID: 214618 Cd Length: 174 Bit Score: 211.74 E-value: 8.84e-64
|
||||||||
| RhoGAP | pfam00620 | RhoGAP domain; GTPase activator proteins towards Rho/Rac/Cdc42-like small GTPases. |
689-835 | 8.35e-59 | ||||
RhoGAP domain; GTPase activator proteins towards Rho/Rac/Cdc42-like small GTPases. Pssm-ID: 459875 Cd Length: 148 Bit Score: 197.00 E-value: 8.35e-59
|
||||||||
| RhoGAP | cd00159 | RhoGAP: GTPase-activator protein (GAP) for Rho-like GTPases; GAPs towards Rho/Rac/Cdc42-like ... |
689-858 | 1.07e-56 | ||||
RhoGAP: GTPase-activator protein (GAP) for Rho-like GTPases; GAPs towards Rho/Rac/Cdc42-like small GTPases. Small GTPases (G proteins) cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when bound to GDP. The Rho family of small G proteins, which includes Cdc42Hs, activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. G proteins generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. The RhoGAPs are one of the major classes of regulators of Rho G proteins. Pssm-ID: 238090 [Multi-domain] Cd Length: 169 Bit Score: 192.13 E-value: 1.07e-56
|
||||||||
| RhoGAP_fRGD1 | cd04398 | RhoGAP_fRGD1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
673-864 | 6.86e-55 | ||||
RhoGAP_fRGD1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of fungal RGD1-like proteins. Yeast Rgd1 is a GAP protein for Rho3 and Rho4 and plays a role in low-pH response. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239863 Cd Length: 192 Bit Score: 188.00 E-value: 6.86e-55
|
||||||||
| RhoGAP_ARHGAP21 | cd04395 | RhoGAP_ARHGAP21: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
687-860 | 3.00e-53 | ||||
RhoGAP_ARHGAP21: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ArhGAP21-like proteins. ArhGAP21 is a multi-domain protein, containing RhoGAP, PH and PDZ domains, and is believed to play a role in the organization of the cell-cell junction complex. It has been shown to function as a GAP of Cdc42 and RhoA, and to interact with alpha-catenin and Arf6. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239860 Cd Length: 196 Bit Score: 183.76 E-value: 3.00e-53
|
||||||||
| RhoGAP_chimaerin | cd04372 | RhoGAP_chimaerin: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
673-864 | 1.29e-48 | ||||
RhoGAP_chimaerin: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of chimaerins. Chimaerins are a family of phorbolester- and diacylglycerol-responsive GAPs specific for the Rho-like GTPase Rac. Chimaerins exist in two alternative splice forms that each contain a C-terminal GAP domain, and a central C1 domain which binds phorbol esters, inducing a conformational change that activates the protein; one splice form is lacking the N-terminal Src homology-2 (SH2) domain. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239837 [Multi-domain] Cd Length: 194 Bit Score: 170.78 E-value: 1.29e-48
|
||||||||
| RhoGAP_Bcr | cd04387 | RhoGAP_Bcr: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of Bcr ... |
673-858 | 1.99e-48 | ||||
RhoGAP_Bcr: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of Bcr (breakpoint cluster region protein)-like proteins. Bcr is a multidomain protein with a variety of enzymatic functions. It contains a RhoGAP and a Rho GEF domain, a Ser/Thr kinase domain, an N-terminal oligomerization domain, and a C-terminal PDZ binding domain, in addition to PH and C2 domains. Bcr is a negative regulator of: i) RacGTPase, via the Rho GAP domain, ii) the Ras-Raf-MEK-ERK pathway, via phosphorylation of the Ras binding protein AF-6, and iii) the Wnt signaling pathway through binding beta-catenin. Bcr can form a complex with beta-catenin and Tcf1. The Wnt signaling pathway is involved in cell proliferation, differentiation, and cell renewal. Bcr was discovered as a fusion partner of Abl. The Bcr-Abl fusion is characteristic for a large majority of chronic myelogenous leukemias (CML). Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239852 [Multi-domain] Cd Length: 196 Bit Score: 170.11 E-value: 1.99e-48
|
||||||||
| PH_ARHGAP9-like | cd13233 | Beta-spectrin pleckstrin homology (PH) domain; ARHGAP family genes encode Rho/Rac/Cdc42-like ... |
477-591 | 3.52e-46 | ||||
Beta-spectrin pleckstrin homology (PH) domain; ARHGAP family genes encode Rho/Rac/Cdc42-like GTPase activating proteins with RhoGAP domain. The ARHGAP members here all have a PH domain upstream of their C-terminal RhoGAP domain. Some have additional N-terminal SH3 and WW domains. The members here include: ARHGAP9, ARHGAP12, ARHGAP15, and ARHGAP27. ARHGAP27 and ARHGAP12 shared the common-domain structure, consisting of SH3, WW, PH, and RhoGAP domains. The PH domain of ArhGAP9 employs a non-canonical phosphoinositide binding mechanism, a variation of the spectrin- Ins(4,5)P2-binding mode, that gives rise to a unique PI binding profile, namely a preference for both PI(4,5)P2 and the PI 3-kinase products PI(3,4,5)P3 and PI(3,4)P2. This lipid binding mechanism is also employed by the PH domain of Tiam1 and Slm1. PH domains have diverse functions, but in general are involved in targeting proteins to the appropriate cellular location or in the interaction with a binding partner. They share little sequence conservation, but all have a common fold, which is electrostatically polarized. Less than 10% of PH domains bind phosphoinositide phosphates (PIPs) with high affinity and specificity. PH domains are distinguished from other PIP-binding domains by their specific high-affinity binding to PIPs with two vicinal phosphate groups: PtdIns(3,4)P2, PtdIns(4,5)P2 or PtdIns(3,4,5)P3 which results in targeting some PH domain proteins to the plasma membrane. A few display strong specificity in lipid binding. Any specificity is usually determined by loop regions or insertions in the N-terminus of the domain, which are not conserved across all PH domains. PH domains are found in cellular signaling proteins such as serine/threonine kinase, tyrosine kinases, regulators of G-proteins, endocytotic GTPases, adaptors, as well as cytoskeletal associated molecules and in lipid associated enzymes. Pssm-ID: 270053 Cd Length: 110 Bit Score: 160.52 E-value: 3.52e-46
|
||||||||
| RhoGAP_Graf | cd04374 | RhoGAP_Graf: GTPase-activator protein (GAP) domain for Rho-like GTPases found in GRAF (GTPase ... |
691-858 | 4.17e-44 | ||||
RhoGAP_Graf: GTPase-activator protein (GAP) domain for Rho-like GTPases found in GRAF (GTPase regulator associated with focal adhesion kinase); Graf is a multi-domain protein, containing SH3 and PH domains, that binds focal adhesion kinase and influences cytoskeletal changes mediated by Rho proteins. Graf exhibits GAP activity toward RhoA and Cdc42, but only weakly activates Rac1. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239839 Cd Length: 203 Bit Score: 158.33 E-value: 4.17e-44
|
||||||||
| RhoGAP_myosin_IX | cd04377 | RhoGAP_myosin_IX: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain ... |
673-859 | 2.53e-41 | ||||
RhoGAP_myosin_IX: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present in class IX myosins. Class IX myosins contain a characteristic head domain, a neck domain, a tail domain which contains a C6H2-zinc binding motif and a RhoGAP domain. Class IX myosins are single-headed, processive myosins that are partly cytoplasmic, and partly associated with membranes and the actin cytoskeleton. Class IX myosins are implicated in the regulation of neuronal morphogenesis and function of sensory systems, like the inner ear. There are two major isoforms, myosin IXA and IXB with several splice variants, which are both expressed in developing neurons. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239842 Cd Length: 186 Bit Score: 149.90 E-value: 2.53e-41
|
||||||||
| RhoGAP_GMIP_PARG1 | cd04378 | RhoGAP_GMIP_PARG1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain ... |
673-858 | 3.94e-39 | ||||
RhoGAP_GMIP_PARG1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of GMIP (Gem interacting protein) and PARG1 (PTPL1-associated RhoGAP1). GMIP plays important roles in neurite growth and axonal guidance, and interacts with Gem, a member of the RGK subfamily of the Ras small GTPase superfamily, through the N-terminal half of the protein. GMIP contains a C-terminal RhoGAP domain. GMIP inhibits RhoA function, but is inactive towards Rac1 and Cdc41. PARG1 interacts with Rap2, also a member of the Ras small GTPase superfamily whose exact function is unknown, and shows strong preference for Rho. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239843 Cd Length: 203 Bit Score: 144.10 E-value: 3.94e-39
|
||||||||
| RhoGAP_p190 | cd04373 | RhoGAP_p190: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
673-859 | 1.50e-36 | ||||
RhoGAP_p190: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of p190-like proteins. p190, also named RhoGAP5, plays a role in neuritogenesis and axon branch stability. p190 shows a preference for Rho, over Rac and Cdc42, and consists of an N-terminal GTPase domain and a C-terminal GAP domain. The central portion of p190 contains important regulatory phosphorylation sites. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239838 Cd Length: 185 Bit Score: 136.05 E-value: 1.50e-36
|
||||||||
| RhoGAP_nadrin | cd04386 | RhoGAP_nadrin: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
671-865 | 4.96e-36 | ||||
RhoGAP_nadrin: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of Nadrin-like proteins. Nadrin, also named Rich-1, has been shown to be involved in the regulation of Ca2+-dependent exocytosis in neurons and recently has been implicated in tight junction maintenance in mammalian epithelium. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239851 Cd Length: 203 Bit Score: 135.28 E-value: 4.96e-36
|
||||||||
| RhoGAP-p50rhoGAP | cd04404 | RhoGAP-p50rhoGAP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
671-864 | 7.22e-36 | ||||
RhoGAP-p50rhoGAP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of p50RhoGAP-like proteins; p50RhoGAP, also known as RhoGAP-1, contains a C-terminal RhoGAP domain and an N-terminal Sec14 domain which binds phosphatidylinositol 3,4,5-trisphosphate (PtdIns(3,4,5)P3). It is ubiquitously expressed and preferentially active on Cdc42. This subgroup also contains closely related ARHGAP8. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239869 [Multi-domain] Cd Length: 195 Bit Score: 134.39 E-value: 7.22e-36
|
||||||||
| RhoGAP_MgcRacGAP | cd04382 | RhoGAP_MgcRacGAP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain ... |
687-857 | 2.29e-35 | ||||
RhoGAP_MgcRacGAP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present in MgcRacGAP proteins. MgcRacGAP plays an important dual role in cytokinesis: i) it is part of centralspindlin-complex, together with the mitotic kinesin MKLP1, which is critical for the structure of the central spindle by promoting microtuble bundling. ii) after phosphorylation by aurora B MgcRacGAP becomes an effective regulator of RhoA and plays an important role in the assembly of the contractile ring and the initiation of cytokinesis. MgcRacGAP-like proteins contain a N-terminal C1-like domain, and a C-terminal RhoGAP domain. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239847 Cd Length: 193 Bit Score: 132.80 E-value: 2.29e-35
|
||||||||
| SH3_ARHGAP27 | cd12069 | Src Homology 3 domain of Rho GTPase-activating protein 27; Rho GTPase-activating proteins ... |
10-66 | 3.54e-33 | ||||
Src Homology 3 domain of Rho GTPase-activating protein 27; Rho GTPase-activating proteins (RhoGAPs or ARHGAPs) bind to Rho proteins and enhance the hydrolysis rates of bound GTP. ARHGAP27, also called CAMGAP1, shows GAP activity towards Rac1 and Cdc42. It binds the adaptor protein CIN85 and may play a role in clathrin-mediated endocytosis. It contains SH3, WW, Pleckstin homology (PH), and RhoGAP domains. SH3 domains bind to proline-rich ligands with moderate affinity and selectivity, preferentially to PxxP motifs; they play a role in the regulation of enzymes by intramolecular interactions, changing the subcellular localization of signal pathway components and mediate multiprotein complex assemblies. Pssm-ID: 213002 Cd Length: 57 Bit Score: 121.85 E-value: 3.54e-33
|
||||||||
| RhoGAP_ARAP | cd04385 | RhoGAP_ARAP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present ... |
685-836 | 7.27e-33 | ||||
RhoGAP_ARAP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present in ARAPs. ARAPs (also known as centaurin deltas) contain, besides the RhoGAP domain, an Arf GAP, ankyrin repeat ras-associating, and PH domains. Since their ArfGAP activity is PIP3-dependent, ARAPs are considered integration points for phosphoinositide, Arf and Rho signaling. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239850 Cd Length: 184 Bit Score: 125.50 E-value: 7.27e-33
|
||||||||
| RhoGAP_CdGAP | cd04384 | RhoGAP_CdGAP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
672-857 | 1.46e-32 | ||||
RhoGAP_CdGAP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of CdGAP-like proteins; CdGAP contains an N-terminal RhoGAP domain and a C-terminal proline-rich region, and it is active on both Cdc42 and Rac1 but not RhoA. CdGAP is recruited to focal adhesions via the interaction with the scaffold protein actopaxin (alpha-parvin). Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239849 [Multi-domain] Cd Length: 195 Bit Score: 124.92 E-value: 1.46e-32
|
||||||||
| RhoGAP_PARG1 | cd04409 | RhoGAP_PARG1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
673-857 | 1.71e-31 | ||||
RhoGAP_PARG1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of PARG1 (PTPL1-associated RhoGAP1). PARG1 was originally cloned as an interaction partner of PTPL1, an intracellular protein-tyrosine phosphatase. PARG1 interacts with Rap2, also a member of the Ras small GTPase superfamily whose exact function is unknown, and shows strong preference for Rho. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239874 Cd Length: 211 Bit Score: 122.61 E-value: 1.71e-31
|
||||||||
| RhoGAP_GMIP | cd04408 | RhoGAP_GMIP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of GMIP ... |
673-858 | 1.54e-30 | ||||
RhoGAP_GMIP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of GMIP (Gem interacting protein). GMIP plays important roles in neurite growth and axonal guidance, and interacts with Gem, a member of the RGK subfamily of the Ras small GTPase superfamily, through the N-terminal half of the protein. GMIP contains a C-terminal RhoGAP domain. GMIP inhibits RhoA function, but is inactive towards Rac1 and Cdc41. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239873 Cd Length: 200 Bit Score: 119.15 E-value: 1.54e-30
|
||||||||
| RhoGAP_ARHGAP18 | cd04391 | RhoGAP_ARHGAP18: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
672-835 | 3.76e-30 | ||||
RhoGAP_ARHGAP18: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ArhGAP18-like proteins. The function of ArhGAP18 is unknown. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239856 Cd Length: 216 Bit Score: 118.60 E-value: 3.76e-30
|
||||||||
| RhoGAP_myosin_IXB | cd04407 | RhoGAP_myosin_IXB: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain ... |
673-859 | 7.17e-30 | ||||
RhoGAP_myosin_IXB: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present in myosins IXB. Class IX myosins contain a characteristic head domain, a neck domain and a tail domain which contains a C6H2-zinc binding motif and a Rho-GAP domain. Class IX myosins are single-headed, processive myosins that are partly cytoplasmic, and partly associated with membranes and the actin cytoskeleton. Class IX myosins are implicated in the regulation of neuronal morphogenesis and function of sensory systems, like the inner ear. There are two major isoforms, myosin IXA and IXB with several splice variants, which are both expressed in developing neurons Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239872 [Multi-domain] Cd Length: 186 Bit Score: 117.01 E-value: 7.17e-30
|
||||||||
| RhoGAP_fBEM3 | cd04400 | RhoGAP_fBEM3: RhoGAP (GTPase-activator [GAP] protein for Rho-like small GTPases) domain of ... |
672-832 | 1.76e-29 | ||||
RhoGAP_fBEM3: RhoGAP (GTPase-activator [GAP] protein for Rho-like small GTPases) domain of fungal BEM3-like proteins. Bem3 is a GAP protein of Cdc42, and is specifically involved in the control of the initial assembly of the septin ring in yeast bud formation. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239865 [Multi-domain] Cd Length: 190 Bit Score: 115.92 E-value: 1.76e-29
|
||||||||
| RhoGAP_SYD1 | cd04379 | RhoGAP_SYD1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present ... |
673-835 | 6.88e-28 | ||||
RhoGAP_SYD1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present in SYD-1_like proteins. Syd-1, first identified and best studied in C.elegans, has been shown to play an important role in neuronal development by specifying axonal properties. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239844 Cd Length: 207 Bit Score: 111.79 E-value: 6.88e-28
|
||||||||
| RhoGAP_ARHGAP22_24_25 | cd04390 | RhoGAP_ARHGAP22_24_25: GTPase-activator protein (GAP) domain for Rho-like GTPases found in ... |
672-864 | 8.01e-28 | ||||
RhoGAP_ARHGAP22_24_25: GTPase-activator protein (GAP) domain for Rho-like GTPases found in ARHGAP22, 24 and 25-like proteins; longer isoforms of these proteins contain an additional N-terminal pleckstrin homology (PH) domain. ARHGAP25 (KIA0053) has been identified as a GAP for Rac1 and Cdc42. Short isoforms (without the PH domain) of ARHGAP24, called RC-GAP72 and p73RhoGAP, and of ARHGAP22, called p68RacGAP, has been shown to be involved in angiogenesis and endothelial cell capillary formation. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239855 [Multi-domain] Cd Length: 199 Bit Score: 111.38 E-value: 8.01e-28
|
||||||||
| SH3_ARHGAP9_like | cd11888 | Src Homology 3 domain of Rho GTPase-activating protein 9 and similar proteins; This subfamily ... |
10-63 | 2.64e-27 | ||||
Src Homology 3 domain of Rho GTPase-activating protein 9 and similar proteins; This subfamily is composed of Rho GTPase-activating proteins including mammalian ARHGAP9, and vertebrate ARHGAPs 12 and 27. RhoGAPs (or ARHGAPs) bind to Rho proteins and enhance the hydrolysis rates of bound GTP. ARHGAP9 functions as a GAP for Rac and Cdc42, but not for RhoA. It negatively regulates cell migration and adhesion. It also acts as a docking protein for the MAP kinases Erk2 and p38alpha, and may facilitate cross-talk between the Rho GTPase and MAPK pathways to control actin remodeling. ARHGAP27, also called CAMGAP1, shows GAP activity towards Rac1 and Cdc42. It binds the adaptor protein CIN85 and may play a role in clathrin-mediated endocytosis. ARHGAP12 has been shown to display GAP activity towards Rac1. It plays a role in regulating HFG-driven cell growth and invasiveness. ARHGAPs in this subfamily contain SH3, WW, Pleckstin homology (PH), and RhoGAP domains. SH3 domains bind to proline-rich ligands with moderate affinity and selectivity, preferentially to PxxP motifs; they play a role in the regulation of enzymes by intramolecular interactions, changing the subcellular localization of signal pathway components and mediate multiprotein complex assemblies. Pssm-ID: 212821 [Multi-domain] Cd Length: 54 Bit Score: 104.76 E-value: 2.64e-27
|
||||||||
| RhoGAP_srGAP | cd04383 | RhoGAP_srGAP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain ... |
688-856 | 7.56e-27 | ||||
RhoGAP_srGAP: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present in srGAPs. srGAPs are components of the intracellular part of Slit-Robo signalling pathway that is important for axon guidance and cell migration. srGAPs contain an N-terminal FCH domain, a central RhoGAP domain and a C-terminal SH3 domain; this SH3 domain interacts with the intracellular proline-rich-tail of the Roundabout receptor (Robo). This interaction with Robo then activates the rhoGAP domain which in turn inhibits Cdc42 activity. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239848 Cd Length: 188 Bit Score: 108.28 E-value: 7.56e-27
|
||||||||
| RhoGAP_myosin_IXA | cd04406 | RhoGAP_myosin_IXA: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain ... |
673-847 | 3.53e-26 | ||||
RhoGAP_myosin_IXA: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present in myosins IXA. Class IX myosins contain a characteristic head domain, a neck domain and a tail domain which contains a C6H2-zinc binding motif and a Rho-GAP domain. Class IX myosins are single-headed, processive myosins that are partly cytoplasmic, and partly associated with membranes and the actin cytoskeleton. Class IX myosins are implicated in the regulation of neuronal morphogenesis and function of sensory systems, like the inner ear. There are two major isoforms, myosin IXA and IXB with several splice variants, which are both expressed in developing neurons. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239871 Cd Length: 186 Bit Score: 106.24 E-value: 3.53e-26
|
||||||||
| RhoGAP_FAM13A1a | cd04393 | RhoGAP_FAM13A1a: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
671-856 | 5.49e-26 | ||||
RhoGAP_FAM13A1a: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of FAM13A1, isoform a-like proteins. The function of FAM13A1a is unknown. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by up several orders of magnitude. Pssm-ID: 239858 [Multi-domain] Cd Length: 189 Bit Score: 106.01 E-value: 5.49e-26
|
||||||||
| RhoGAP_ARHGAP6 | cd04376 | RhoGAP_ARHGAP6: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
688-834 | 8.43e-24 | ||||
RhoGAP_ARHGAP6: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ArhGAP6-like proteins. ArhGAP6 shows GAP activity towards RhoA, but not towards Cdc42 and Rac1. ArhGAP6 is often deleted in microphthalmia with linear skin defects syndrome (MLS); MLS is a severe X-linked developmental disorder. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239841 Cd Length: 206 Bit Score: 100.21 E-value: 8.43e-24
|
||||||||
| RhoGAP_ARHGAP20 | cd04402 | RhoGAP_ARHGAP20: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
673-864 | 4.78e-23 | ||||
RhoGAP_ARHGAP20: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ArhGAP20-like proteins. ArhGAP20, also known as KIAA1391 and RA-RhoGAP, contains a RhoGAP, a RA, and a PH domain, and ANXL repeats. ArhGAP20 is activated by Rap1 and induces inactivation of Rho, which in turn leads to neurite outgrowth. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239867 Cd Length: 192 Bit Score: 97.37 E-value: 4.78e-23
|
||||||||
| RhoGap_RalBP1 | cd04381 | RhoGap_RalBP1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain ... |
673-832 | 1.38e-22 | ||||
RhoGap_RalBP1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present in RalBP1 proteins, also known as RLIP, RLIP76 or cytocentrin. RalBP1 plays an important role in endocytosis during interphase. During mitosis, RalBP1 transiently associates with the centromere and has been shown to play an essential role in the proper assembly of the mitotic apparatus. RalBP1 is an effector of the Ral GTPase which itself is an effector of Ras. RalBP1 contains a RhoGAP domain, which shows weak activity towards Rac1 and Cdc42, but not towards Ral, and a Ral effector domain binding motif. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239846 [Multi-domain] Cd Length: 182 Bit Score: 95.97 E-value: 1.38e-22
|
||||||||
| RhoGAP-ARHGAP11A | cd04394 | RhoGAP-ARHGAP11A: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
672-863 | 1.92e-20 | ||||
RhoGAP-ARHGAP11A: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ArhGAP11A-like proteins. The mouse homolog of human ArhGAP11A has been detected as a gene exclusively expressed in immature ganglion cells, potentially playing a role in retinal development. The exact function of ArhGAP11A is unknown. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239859 [Multi-domain] Cd Length: 202 Bit Score: 90.22 E-value: 1.92e-20
|
||||||||
| RhoGAP_fLRG1 | cd04397 | RhoGAP_fLRG1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
673-842 | 1.71e-19 | ||||
RhoGAP_fLRG1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of fungal LRG1-like proteins. Yeast Lrg1p is required for efficient cell fusion, and mother-daughter cell separation, possibly through acting as a RhoGAP specifically regulating 1,3-beta-glucan synthesis. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239862 Cd Length: 213 Bit Score: 87.81 E-value: 1.71e-19
|
||||||||
| RhoGAP_KIAA1688 | cd04389 | RhoGAP_KIAA1688: GTPase-activator protein (GAP) domain for Rho-like GTPases found in ... |
673-854 | 2.56e-17 | ||||
RhoGAP_KIAA1688: GTPase-activator protein (GAP) domain for Rho-like GTPases found in KIAA1688-like proteins; KIAA1688 is a protein of unknown function that contains a RhoGAP domain and a myosin tail homology 4 (MyTH4) domain. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239854 Cd Length: 187 Bit Score: 80.90 E-value: 2.56e-17
|
||||||||
| SH3_ARHGAP12 | cd12070 | Src Homology 3 domain of Rho GTPase-activating protein 12; Rho GTPase-activating proteins ... |
9-67 | 1.11e-16 | ||||
Src Homology 3 domain of Rho GTPase-activating protein 12; Rho GTPase-activating proteins (RhoGAPs or ARHGAPs) bind to Rho proteins and enhance the hydrolysis rates of bound GTP. ARHGAP12 has been shown to display GAP activity towards Rac1. It plays a role in regulating hepatocyte growth factor (HGF)-driven cell growth and invasiveness. It contains SH3, WW, Pleckstin homology (PH), and RhoGAP domains. SH3 domains bind to proline-rich ligands with moderate affinity and selectivity, preferentially to PxxP motifs; they play a role in the regulation of enzymes by intramolecular interactions, changing the subcellular localization of signal pathway components and mediate multiprotein complex assemblies. Pssm-ID: 213003 Cd Length: 60 Bit Score: 75.01 E-value: 1.11e-16
|
||||||||
| RhoGAP_DLC1 | cd04375 | RhoGAP_DLC1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
672-832 | 6.16e-16 | ||||
RhoGAP_DLC1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of DLC1-like proteins. DLC1 shows in vitro GAP activity towards RhoA and CDC42. Beside its C-terminal GAP domain, DLC1 also contains a SAM (sterile alpha motif) and a START (StAR-related lipid transfer action) domain. DLC1 has tumor suppressor activity in cell culture. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239840 Cd Length: 220 Bit Score: 77.84 E-value: 6.16e-16
|
||||||||
| RhoGAP_ARHGAP19 | cd04392 | RhoGAP_ARHGAP19: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
704-835 | 8.52e-16 | ||||
RhoGAP_ARHGAP19: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ArhGAP19-like proteins. The function of ArhGAP19 is unknown. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239857 Cd Length: 208 Bit Score: 77.12 E-value: 8.52e-16
|
||||||||
| PH_beta_spectrin | cd10571 | Beta-spectrin pleckstrin homology (PH) domain; Beta spectrin binds actin and functions as a ... |
479-586 | 6.65e-15 | ||||
Beta-spectrin pleckstrin homology (PH) domain; Beta spectrin binds actin and functions as a major component of the cytoskeleton underlying cellular membranes. Beta spectrin consists of multiple spectrin repeats followed by a PH domain, which binds to inositol-1,4,5-trisphosphate. The PH domain of beta-spectrin is thought to play a role in the association of spectrin with the plasma membrane of cells. PH domains have diverse functions, but in general are involved in targeting proteins to the appropriate cellular location or in the interaction with a binding partner. They share little sequence conservation, but all have a common fold, which is electrostatically polarized. Less than 10% of PH domains bind phosphoinositide phosphates (PIPs) with high affinity and specificity. PH domains are distinguished from other PIP-binding domains by their specific high-affinity binding to PIPs with two vicinal phosphate groups: PtdIns(3,4)P2, PtdIns(4,5)P2 or PtdIns(3,4,5)P3 which results in targeting some PH domain proteins to the plasma membrane. A few display strong specificity in lipid binding. Any specificity is usually determined by loop regions or insertions in the N-terminus of the domain, which are not conserved across all PH domains. PH domains are found in cellular signaling proteins such as serine/threonine kinase, tyrosine kinases, regulators of G-proteins, endocytotic GTPases, adaptors, as well as cytoskeletal associated molecules and in lipid associated enzymes. Pssm-ID: 269975 Cd Length: 106 Bit Score: 71.11 E-value: 6.65e-15
|
||||||||
| RhoGAP_p85 | cd04388 | RhoGAP_p85: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present ... |
689-866 | 2.07e-11 | ||||
RhoGAP_p85: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present in the p85 isoforms of the regulatory subunit of the class IA PI3K (phosphatidylinositol 3'-kinase). This domain is also called Bcr (breakpoint cluster region protein) homology (BH) domain. Class IA PI3Ks are heterodimers, containing a regulatory subunit (p85) and a catalytic subunit (p110) and are activated by growth factor receptor tyrosine kinases (RTKs); this activation is mediated by the p85 subunit. p85 isoforms, alpha and beta, contain a C-terminal p110-binding domain flanked by two SH2 domains, an N-terminal SH3 domain, and a RhoGAP domain flanked by two proline-rich regions. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239853 Cd Length: 200 Bit Score: 64.12 E-value: 2.07e-11
|
||||||||
| RhoGAP_fSAC7_BAG7 | cd04396 | RhoGAP_fSAC7_BAG7: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain ... |
687-864 | 2.52e-10 | ||||
RhoGAP_fSAC7_BAG7: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of fungal SAC7 and BAG7-like proteins. Both proteins are GTPase activating proteins of Rho1, but differ functionally in vivo: SAC7, but not BAG7, is involved in the control of Rho1-mediated activation of the PKC-MPK1 pathway. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239861 Cd Length: 225 Bit Score: 61.27 E-value: 2.52e-10
|
||||||||
| PH_ARHGAP21-like | cd01253 | ARHGAP21 and related proteins pleckstrin homology (PH) domain; ARHGAP family genes encode Rho ... |
477-585 | 2.81e-10 | ||||
ARHGAP21 and related proteins pleckstrin homology (PH) domain; ARHGAP family genes encode Rho/Rac/Cdc42-like GTPase activating proteins with a RhoGAP domain. These proteins functions as a GTPase-activating protein (GAP) for RHOA and CDC42. ARHGAP21 controls the Arp2/3 complex and F-actin dynamics at the Golgi complex by regulating the activity of the small GTPase Cdc42. It is recruited to the Golgi by to GTPase, ARF1, through its PH domain and its helical motif. It is also required for CTNNA1 recruitment to adherens junctions. ARHGAP21 and it related proteins all contains a PH domain and a RhoGAP domain. Some of the members have additional N-terminal domains including PDZ, SH3, and SPEC. The ARHGAP21 PH domain interacts with the GTPbound forms of both ARF1 and ARF6 ARF-binding domain/ArfBD. The members here include: ARHGAP15, ARHGAP21, and ARHGAP23. PH domains have diverse functions, but in general are involved in targeting proteins to the appropriate cellular location or in the interaction with a binding partner. They share little sequence conservation, but all have a common fold, which is electrostatically polarized. Less than 10% of PH domains bind phosphoinositide phosphates (PIPs) with high affinity and specificity. PH domains are distinguished from other PIP-binding domains by their specific high-affinity binding to PIPs with two vicinal phosphate groups: PtdIns(3,4)P2, PtdIns(4,5)P2 or PtdIns(3,4,5)P3 which results in targeting some PH domain proteins to the plasma membrane. A few display strong specificity in lipid binding. Any specificity is usually determined by loop regions or insertions in the N-terminus of the domain, which are not conserved across all PH domains. PH domains are found in cellular signaling proteins such as serine/threonine kinase, tyrosine kinases, regulators of G-proteins, endocytotic GTPases, adaptors, as well as cytoskeletal associated molecules and in lipid associated enzymes. Pssm-ID: 269955 Cd Length: 113 Bit Score: 58.15 E-value: 2.81e-10
|
||||||||
| SH3_ARHGAP9 | cd12143 | Src Homology 3 domain of Rho GTPase-activating protein 9 and similar proteins; Rho ... |
17-63 | 6.74e-10 | ||||
Src Homology 3 domain of Rho GTPase-activating protein 9 and similar proteins; Rho GTPase-activating proteins (RhoGAPs or ARHGAPs) bind to Rho proteins and enhance the hydrolysis rates of bound GTP. ARHGAP9 functions as a GAP for Rac and Cdc42, but not for RhoA. It negatively regulates cell migration and adhesion. It also acts as a docking protein for the MAP kinases Erk2 and p38alpha, and may facilitate cross-talk between the Rho GTPase and MAPK pathways to control actin remodeling. It contains SH3, WW, Pleckstin homology (PH), and RhoGAP domains. SH3 domains bind to proline-rich ligands with moderate affinity and selectivity, preferentially to PxxP motifs; they play a role in the regulation of enzymes by intramolecular interactions, changing the subcellular localization of signal pathway components and mediate multiprotein complex assemblies. Pssm-ID: 213019 Cd Length: 57 Bit Score: 55.31 E-value: 6.74e-10
|
||||||||
| PH | smart00233 | Pleckstrin homology domain; Domain commonly found in eukaryotic signalling proteins. The ... |
475-590 | 2.52e-09 | ||||
Pleckstrin homology domain; Domain commonly found in eukaryotic signalling proteins. The domain family possesses multiple functions including the abilities to bind inositol phosphates, and various proteins. PH domains have been found to possess inserted domains (such as in PLC gamma, syntrophins) and to be inserted within other domains. Mutations in Brutons tyrosine kinase (Btk) within its PH domain cause X-linked agammaglobulinaemia (XLA) in patients. Point mutations cluster into the positively charged end of the molecule around the predicted binding site for phosphatidylinositol lipids. Pssm-ID: 214574 [Multi-domain] Cd Length: 102 Bit Score: 55.25 E-value: 2.52e-09
|
||||||||
| RhoGAP_OCRL1 | cd04380 | RhoGAP_OCRL1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain ... |
740-839 | 2.66e-08 | ||||
RhoGAP_OCRL1: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain present in OCRL1-like proteins. OCRL1 (oculocerebrorenal syndrome of Lowe 1)-like proteins contain two conserved domains: a central inositol polyphosphate 5-phosphatase domain and a C-terminal Rho GAP domain, this GAP domain lacks the catalytic residue and therefore maybe inactive. OCRL-like proteins are type II inositol polyphosphate 5-phosphatases that can hydrolyze lipid PI(4,5)P2 and PI(3,4,5)P3 and soluble Ins(1,4,5)P3 and Ins(1,3,4,5)P4, but their individual specificities vary. The functionality of the RhoGAP domain is still unclear. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239845 Cd Length: 220 Bit Score: 55.42 E-value: 2.66e-08
|
||||||||
| WW | pfam00397 | WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds ... |
247-278 | 1.71e-07 | ||||
WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds proline-rich peptide motifs in vitro. Pssm-ID: 459800 [Multi-domain] Cd Length: 30 Bit Score: 47.88 E-value: 1.71e-07
|
||||||||
| RhoGAP_fRGD2 | cd04399 | RhoGAP_fRGD2: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of ... |
673-866 | 2.04e-07 | ||||
RhoGAP_fRGD2: RhoGAP (GTPase-activator protein [GAP] for Rho-like small GTPases) domain of fungal RGD2-like proteins. Yeast Rgd2 is a GAP protein for Cdc42 and Rho5. Small GTPases cluster into distinct families, and all act as molecular switches, active in their GTP-bound form but inactive when GDP-bound. The Rho family of GTPases activates effectors involved in a wide variety of developmental processes, including regulation of cytoskeleton formation, cell proliferation and the JNK signaling pathway. GTPases generally have a low intrinsic GTPase hydrolytic activity but there are family-specific groups of GAPs that enhance the rate of GTP hydrolysis by several orders of magnitude. Pssm-ID: 239864 Cd Length: 212 Bit Score: 52.33 E-value: 2.04e-07
|
||||||||
| WW | smart00456 | Domain with 2 conserved Trp (W) residues; Also known as the WWP or rsp5 domain. Binds ... |
246-278 | 2.40e-07 | ||||
Domain with 2 conserved Trp (W) residues; Also known as the WWP or rsp5 domain. Binds proline-rich polypeptides. Pssm-ID: 197736 [Multi-domain] Cd Length: 33 Bit Score: 47.60 E-value: 2.40e-07
|
||||||||
| SH3_ITK | cd11908 | Src Homology 3 domain of Interleukin-2-inducible T-cell Kinase; ITK (also known as Tsk or Emt) ... |
12-66 | 1.86e-06 | ||||
Src Homology 3 domain of Interleukin-2-inducible T-cell Kinase; ITK (also known as Tsk or Emt) is a cytoplasmic (or nonreceptor) tyr kinase containing Src homology protein interaction domains (SH3, SH2) N-terminal to the catalytic tyr kinase domain. It also contains an N-terminal pleckstrin homology (PH) domain, which binds the products of PI3K and allows membrane recruitment and activation, and the Tec homology (TH) domain, which contains proline-rich and zinc-binding regions. ITK is expressed in T-cells and mast cells, and is important in their development and differentiation. Of the three Tec kinases expressed in T-cells, ITK plays the predominant role in T-cell receptor (TCR) signaling. It is activated by phosphorylation upon TCR crosslinking and is involved in the pathway resulting in phospholipase C-gamma1 activation and actin polymerization. It also plays a role in the downstream signaling of the T-cell costimulatory receptor CD28, the T-cell surface receptor CD2, and the chemokine receptor CXCR4. In addition, ITK is crucial for the development of T-helper(Th)2 effector responses. SH3 domains are protein interaction domains that bind to proline-rich ligands with moderate affinity and selectivity, preferentially to PxxP motifs. They play versatile and diverse roles in the cell including the regulation of enzymes, changing the subcellular localization of signaling pathway components, and mediating the formation of multiprotein complex assemblies. Pssm-ID: 212841 [Multi-domain] Cd Length: 56 Bit Score: 45.78 E-value: 1.86e-06
|
||||||||
| WW | cd00201 | Two conserved tryptophans domain; also known as the WWP or rsp5 domain; around 40 amino acids; ... |
250-278 | 1.98e-06 | ||||
Two conserved tryptophans domain; also known as the WWP or rsp5 domain; around 40 amino acids; functions as an interaction module in a diverse set of signalling proteins; binds specific proline-rich sequences but at low affinities compared to other peptide recognition proteins such as antibodies and receptors; WW domains have a single groove formed by a conserved Trp and Tyr which recognizes a pair of residues of the sequence X-Pro; variable loops and neighboring domains confer specificity in this domain; there are five distinct groups based on binding: 1) PPXY motifs 2) the PPLP motif; 3) PGM motifs; 4) PSP or PTP motifs; 5) PR motifs. Pssm-ID: 238122 [Multi-domain] Cd Length: 31 Bit Score: 44.83 E-value: 1.98e-06
|
||||||||
| SH3_9 | pfam14604 | Variant SH3 domain; |
13-65 | 8.16e-06 | ||||
Variant SH3 domain; Pssm-ID: 434066 [Multi-domain] Cd Length: 49 Bit Score: 43.76 E-value: 8.16e-06
|
||||||||
| PH | cd00821 | Pleckstrin homology (PH) domain; PH domains have diverse functions, but in general are ... |
477-585 | 8.95e-06 | ||||
Pleckstrin homology (PH) domain; PH domains have diverse functions, but in general are involved in targeting proteins to the appropriate cellular location or in the interaction with a binding partner. They share little sequence conservation, but all have a common fold, which is electrostatically polarized. Less than 10% of PH domains bind phosphoinositide phosphates (PIPs) with high affinity and specificity. PH domains are distinguished from other PIP-binding domains by their specific high-affinity binding to PIPs with two vicinal phosphate groups: PtdIns(3,4)P2, PtdIns(4,5)P2 or PtdIns(3,4,5)P3 which results in targeting some PH domain proteins to the plasma membrane. A few display strong specificity in lipid binding. Any specificity is usually determined by loop regions or insertions in the N-terminus of the domain, which are not conserved across all PH domains. PH domains are found in cellular signaling proteins such as serine/threonine kinase, tyrosine kinases, regulators of G-proteins, endocytotic GTPases, adaptors, as well as cytoskeletal associated molecules and in lipid associated enzymes. Pssm-ID: 275388 [Multi-domain] Cd Length: 92 Bit Score: 44.84 E-value: 8.95e-06
|
||||||||
| SH3_Tec_like | cd11768 | Src Homology 3 domain of Tec-like Protein Tyrosine Kinases; The Tec (Tyrosine kinase expressed ... |
17-66 | 1.80e-05 | ||||
Src Homology 3 domain of Tec-like Protein Tyrosine Kinases; The Tec (Tyrosine kinase expressed in hepatocellular carcinoma) subfamily is composed of Tec, Btk, Bmx (Etk), Itk (Tsk, Emt), Rlk (Txk), and similar proteins. They are cytoplasmic (or nonreceptor) tyr kinases containing Src homology protein interaction domains (SH3, SH2) N-terminal to the catalytic tyr kinase domain. Most Tec subfamily members (except Rlk) also contain an N-terminal pleckstrin homology (PH) domain, which binds the products of PI3K and allows membrane recruitment and activation. In addition, some members contain the Tec homology (TH) domain, which contains proline-rich and zinc-binding regions. Tec kinases are expressed mainly by haematopoietic cells, although Tec and Bmx are also found in endothelial cells. B-cells express Btk and Tec, while T-cells express Itk, Txk, and Tec. Collectively, Tec kinases are expressed in a variety of myeloid cells such as mast cells, platelets, macrophages, and dendritic cells. Each Tec kinase shows a distinct cell-type pattern of expression. The function of Tec kinases in lymphoid cells have been studied extensively. They play important roles in the development, differentiation, maturation, regulation, survival, and function of B-cells and T-cells. SH3 domains are protein interaction domains that bind to proline-rich ligands with moderate affinity and selectivity, preferentially to PxxP motifs. They play versatile and diverse roles in the cell including the regulation of enzymes, changing the subcellular localization of signaling pathway components, and mediating the formation of multiprotein complex assemblies. Pssm-ID: 212702 [Multi-domain] Cd Length: 54 Bit Score: 42.65 E-value: 1.80e-05
|
||||||||
| SH3_SPIN90 | cd11849 | Src homology 3 domain of SH3 protein interacting with Nck, 90 kDa (SPIN90); SPIN90 is also ... |
17-65 | 2.53e-05 | ||||
Src homology 3 domain of SH3 protein interacting with Nck, 90 kDa (SPIN90); SPIN90 is also called NCK interacting protein with SH3 domain (NCKIPSD), Dia-interacting protein (DIP), 54 kDa vimentin-interacting protein (VIP54), or WASP-interacting SH3-domain protein (WISH). It is an F-actin binding protein that regulates actin polymerization and endocytosis. It associates with the Arp2/3 complex near actin filaments and determines filament localization at the leading edge of lamellipodia. SPIN90 is expressed in the early stages of neuronal differentiation and plays a role in regulating growth cone dynamics and neurite outgrowth. It also interacts with IRSp53 and regulates cell motility by playing a role in the formation of membrane protrusions. SPIN90 contains an N-terminal SH3 domain, a proline-rich domain, and a C-terminal VCA (verprolin-homology and cofilin-like acidic) domain. SH3 domains are protein interaction domains that bind to proline-rich ligands with moderate affinity and selectivity, preferentially to PxxP motifs. They play versatile and diverse roles in the cell including the regulation of enzymes, changing the subcellular localization of signaling pathway components, and mediating the formation of multiprotein complex assemblies. Pssm-ID: 212783 [Multi-domain] Cd Length: 53 Bit Score: 42.30 E-value: 2.53e-05
|
||||||||
| PH | pfam00169 | PH domain; PH stands for pleckstrin homology. |
477-589 | 2.75e-05 | ||||
PH domain; PH stands for pleckstrin homology. Pssm-ID: 459697 [Multi-domain] Cd Length: 105 Bit Score: 43.71 E-value: 2.75e-05
|
||||||||
| PRP40 | COG5104 | Splicing factor [RNA processing and modification]; |
239-376 | 5.16e-05 | ||||
Splicing factor [RNA processing and modification]; Pssm-ID: 227435 [Multi-domain] Cd Length: 590 Bit Score: 47.00 E-value: 5.16e-05
|
||||||||
| SH3_Nck_1 | cd11765 | First Src Homology 3 domain of Nck adaptor proteins; Nck adaptor proteins regulate actin ... |
10-64 | 8.12e-05 | ||||
First Src Homology 3 domain of Nck adaptor proteins; Nck adaptor proteins regulate actin cytoskeleton dynamics by linking proline-rich effector molecules to protein tyrosine kinases and phosphorylated signaling intermediates. They contain three SH3 domains and a C-terminal SH2 domain. They function downstream of the PDGFbeta receptor and are involved in Rho GTPase signaling and actin dynamics. Vertebrates contain two Nck adaptor proteins: Nck1 (also called Nckalpha) and Nck2 (also called Nckbeta or Growth factor receptor-bound protein 4, Grb4), which show partly overlapping functions but also bind distinct targets. Their SH3 domains are involved in recruiting downstream effector molecules, such as the N-WASP/Arp2/3 complex, which when activated induces actin polymerization that results in the production of pedestals, or protrusions of the plasma membrane. The first SH3 domain of Nck proteins preferentially binds the PxxDY sequence, which is present in the CD3e cytoplasmic tail. This binding inhibits phosphorylation by Src kinases, resulting in the downregulation of TCR surface expression. SH3 domains are protein interaction domains that usually bind to proline-rich ligands with moderate affinity and selectivity, preferentially a PxxP motif. They play versatile and diverse roles in the cell including the regulation of enzymes, changing the subcellular localization of signaling pathway components, and mediating the formation of multiprotein complex assemblies. Pssm-ID: 212699 [Multi-domain] Cd Length: 51 Bit Score: 40.86 E-value: 8.12e-05
|
||||||||
| PH1_Pleckstrin_2 | cd13301 | Pleckstrin 2 Pleckstrin homology (PH) domain, repeat 1; Pleckstrin is a protein found in ... |
498-594 | 2.03e-04 | ||||
Pleckstrin 2 Pleckstrin homology (PH) domain, repeat 1; Pleckstrin is a protein found in platelets. This name is derived from platelet and leukocyte C kinase substrate and the KSTR string of amino acids. Pleckstrin 2 contains two PH domains and a DEP (dishvelled, egl-10, and pleckstrin) domain. Unlike pleckstrin 1, pleckstrin 2 does not contain obvious sites of PKC phosphorylation. Pleckstrin 2 plays a role in actin rearrangement, large lamellipodia and peripheral ruffle formation, and may help orchestrate cytoskeletal arrangement. The PH domains of pleckstrin 2 are thought to contribute to lamellipodia formation. This cd contains the first PH domain repeat. PH domains have diverse functions, but in general are involved in targeting proteins to the appropriate cellular location or in the interaction with a binding partner. They share little sequence conservation, but all have a common fold, which is electrostatically polarized. Less than 10% of PH domains bind phosphoinositide phosphates (PIPs) with high affinity and specificity. PH domains are distinguished from other PIP-binding domains by their specific high-affinity binding to PIPs with two vicinal phosphate groups: PtdIns(3,4)P2, PtdIns(4,5)P2 or PtdIns(3,4,5)P3 which results in targeting some PH domain proteins to the plasma membrane. A few display strong specificity in lipid binding. Any specificity is usually determined by loop regions or insertions in the N-terminus of the domain, which are not conserved across all PH domains. PH domains are found in cellular signaling proteins such as serine/threonine kinase, tyrosine kinases, regulators of G-proteins, endocytotic GTPases, adaptors, as well as cytoskeletal associated molecules and in lipid associated enzymes. Pssm-ID: 270113 Cd Length: 108 Bit Score: 41.59 E-value: 2.03e-04
|
||||||||
| PH_EFA6 | cd13295 | Exchange Factor for ARF6 Pleckstrin homology (PH) domain; EFA6 (also called PSD/pleckstrin and ... |
477-598 | 2.25e-04 | ||||
Exchange Factor for ARF6 Pleckstrin homology (PH) domain; EFA6 (also called PSD/pleckstrin and Sec7 domain containing) is an guanine nucleotide exchange factor for ADP ribosylation factor 6 (ARF6), which is involved in membrane recycling. EFA6 has four structurally related polypeptides: EFA6A, EFA6B, EFA6C and EFA6D. It consists of a N-terminal proline rich region (PR), a SEC7 domain, a PH domain, a PR, a coiled-coil region, and a C-terminal PR. The EFA6 PH domain regulates its association with the plasma membrane. EFA6 activates Arf6 through its Sec7 catalytic domain and modulates this activity through its C-terminal domain, which rearranges the actin cytoskeleton in fibroblastic cell lines. PH domains have diverse functions, but in general are involved in targeting proteins to the appropriate cellular location or in the interaction with a binding partner. They share little sequence conservation, but all have a common fold, which is electrostatically polarized. Less than 10% of PH domains bind phosphoinositide phosphates (PIPs) with high affinity and specificity. PH domains are distinguished from other PIP-binding domains by their specific high-affinity binding to PIPs with two vicinal phosphate groups: PtdIns(3,4)P2, PtdIns(4,5)P2 or PtdIns(3,4,5)P3 which results in targeting some PH domain proteins to the plasma membrane. A few display strong specificity in lipid binding. Any specificity is usually determined by loop regions or insertions in the N-terminus of the domain, which are not conserved across all PH domains. PH domains are found in cellular signaling proteins such as serine/threonine kinase, tyrosine kinases, regulators of G-proteins, endocytotic GTPases, adaptors, as well as cytoskeletal associated molecules and in lipid associated enzymes. Pssm-ID: 270107 Cd Length: 126 Bit Score: 41.93 E-value: 2.25e-04
|
||||||||
| PH1_Tiam1_2 | cd01230 | T-lymphoma invasion and metastasis 1 and 2 Pleckstrin Homology (PH) domain, N-terminal domain; ... |
477-585 | 4.00e-04 | ||||
T-lymphoma invasion and metastasis 1 and 2 Pleckstrin Homology (PH) domain, N-terminal domain; Tiam1 activates Rac GTPases to induce membrane ruffling and cell motility while Tiam2 (also called STEF (SIF (still life) and Tiam1 like-exchange factor) contributes to neurite growth. Tiam1/2 are Dbl-family of GEFs that possess a Dbl(DH) domain with a PH domain in tandem. DH-PH domain catalyzes the GDP/GTP exchange reaction in the GTPase cycle and facillitating the switch between inactive GDP-bound and active GTP-bound states. Tiam1/2 possess two PH domains, which are often referred to as PHn and PHc domains. The DH-PH tandem domain is made up of the PHc domain while the PHn is part of a novel N-terminal PHCCEx domain which is made up of the PHn domain, a coiled coil region(CC), and an extra region (Ex). PHCCEx mediates binding to plasma membranes and signalling proteins in the activation of Rac GTPases. The PH domain resembles the beta-spectrin PH domain, suggesting non-canonical phosphatidylinositol binding. CC and Ex form a positively charged surface for protein binding. There are 2 motifs in Tiam1/2-interacting proteins that bind to the PHCCEx domain: Motif-I in CD44, ephrinBs, and the NMDA receptor and Motif-II in Par3 and JIP2.Neither of these fall in the PHn domain. PH domains have diverse functions, but in general are involved in targeting proteins to the appropriate cellular location or in the interaction with a binding partner. They share little sequence conservation, but all have a common fold, which is electrostatically polarized. Less than 10% of PH domains bind phosphoinositide phosphates (PIPs) with high affinity and specificity. PH domains are distinguished from other PIP-binding domains by their specific high-affinity binding to PIPs with two vicinal phosphate groups: PtdIns(3,4)P2, PtdIns(4,5)P2 or PtdIns(3,4,5)P3 which results in targeting some PH domain proteins to the plasma membrane. A few display strong specificity in lipid binding. Any specificity is usually determined by loop regions or insertions in the N-terminus of the domain, which are not conserved across all PH domains. PH domains are found in cellular signaling proteins such as serine/threonine kinase, tyrosine kinases, regulators of G-proteins, endocytotic GTPases, adaptors, as well as cytoskeletal associated molecules and in lipid associated enzymes. Pssm-ID: 269937 Cd Length: 127 Bit Score: 41.29 E-value: 4.00e-04
|
||||||||
| PH_AtPH1 | cd13276 | Arabidopsis thaliana Pleckstrin homolog (PH) 1 (AtPH1) PH domain; AtPH1 is expressed in all ... |
496-593 | 9.27e-04 | ||||
Arabidopsis thaliana Pleckstrin homolog (PH) 1 (AtPH1) PH domain; AtPH1 is expressed in all plant tissue and is proposed to be the plant homolog of human pleckstrin. Pleckstrin consists of two PH domains separated by a linker region, while AtPH has a single PH domain with a short N-terminal extension. AtPH1 binds PtdIns3P specifically and is thought to be an adaptor molecule since it has no obvious catalytic functions. PH domains have diverse functions, but in general are involved in targeting proteins to the appropriate cellular location or in the interaction with a binding partner. They share little sequence conservation, but all have a common fold, which is electrostatically polarized. Less than 10% of PH domains bind phosphoinositide phosphates (PIPs) with high affinity and specificity. PH domains are distinguished from other PIP-binding domains by their specific high-affinity binding to PIPs with two vicinal phosphate groups: PtdIns(3,4)P2, PtdIns(4,5)P2 or PtdIns(3,4,5)P3 which results in targeting some PH domain proteins to the plasma membrane. A few display strong specificity in lipid binding. Any specificity is usually determined by loop regions or insertions in the N-terminus of the domain, which are not conserved across all PH domains. PH domains are found in cellular signaling proteins such as serine/threonine kinase, tyrosine kinases, regulators of G-proteins, endocytotic GTPases, adaptors, as well as cytoskeletal associated molecules and in lipid associated enzymes. Pssm-ID: 270095 Cd Length: 106 Bit Score: 39.61 E-value: 9.27e-04
|
||||||||
| WW | pfam00397 | WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds ... |
415-442 | 1.71e-03 | ||||
WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds proline-rich peptide motifs in vitro. Pssm-ID: 459800 [Multi-domain] Cd Length: 30 Bit Score: 36.71 E-value: 1.71e-03
|
||||||||
| WW | smart00456 | Domain with 2 conserved Trp (W) residues; Also known as the WWP or rsp5 domain. Binds ... |
417-443 | 2.69e-03 | ||||
Domain with 2 conserved Trp (W) residues; Also known as the WWP or rsp5 domain. Binds proline-rich polypeptides. Pssm-ID: 197736 [Multi-domain] Cd Length: 33 Bit Score: 36.04 E-value: 2.69e-03
|
||||||||
| SH3_BTK | cd11906 | Src Homology 3 domain of Bruton's tyrosine kinase; BTK is a cytoplasmic (or nonreceptor) tyr ... |
17-66 | 6.51e-03 | ||||
Src Homology 3 domain of Bruton's tyrosine kinase; BTK is a cytoplasmic (or nonreceptor) tyr kinase containing Src homology protein interaction domains (SH3, SH2) N-terminal to the catalytic tyr kinase domain. It also contains an N-terminal pleckstrin homology (PH) domain, which binds the products of PI3K and allows membrane recruitment and activation, and the Tec homology (TH) domain with proline-rich and zinc-binding regions. Btk is expressed in B-cells, and a variety of myeloid cells including mast cells, platelets, neutrophils, and dendrictic cells. It interacts with a variety of partners, from cytosolic proteins to nuclear transcription factors, suggesting a diversity of functions. Stimulation of a diverse array of cell surface receptors, including antigen engagement of the B-cell receptor (BCR), leads to PH-mediated membrane translocation of Btk and subsequent phosphorylation by Src kinase and activation. Btk plays an important role in the life cycle of B-cells including their development, differentiation, proliferation, survival, and apoptosis. Mutations in Btk cause the primary immunodeficiency disease, X-linked agammaglobulinaemia (XLA) in humans. SH3 domains are protein interaction domains that bind to proline-rich ligands with moderate affinity and selectivity, preferentially to PxxP motifs. They play versatile and diverse roles in the cell including the regulation of enzymes, changing the subcellular localization of signaling pathway components, and mediating the formation of multiprotein complex assemblies. Pssm-ID: 212839 [Multi-domain] Cd Length: 55 Bit Score: 35.57 E-value: 6.51e-03
|
||||||||
| WW | pfam00397 | WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds ... |
301-328 | 7.06e-03 | ||||
WW domain; The WW domain is a protein module with two highly conserved tryptophans that binds proline-rich peptide motifs in vitro. Pssm-ID: 459800 [Multi-domain] Cd Length: 30 Bit Score: 34.79 E-value: 7.06e-03
|
||||||||
| SH3_Nck2_1 | cd11899 | First Src Homology 3 domain of Nck2 adaptor protein; Nck2 (also called Nckbeta or Growth ... |
11-64 | 9.20e-03 | ||||
First Src Homology 3 domain of Nck2 adaptor protein; Nck2 (also called Nckbeta or Growth factor receptor-bound protein 4, Grb4) plays a crucial role in connecting signaling pathways of tyrosine kinase receptors and important effectors in actin dynamics and cytoskeletal remodeling. It binds neuronal signaling proteins such as ephrinB and Disabled-1 (Dab-1) exclusively. Nck adaptor proteins regulate actin cytoskeleton dynamics by linking proline-rich effector molecules to protein tyrosine kinases and phosphorylated signaling intermediates. They contain three SH3 domains and a C-terminal SH2 domain. They function downstream of the PDGFbeta receptor and are involved in Rho GTPase signaling and actin dynamics. Vertebrates contain two Nck adaptor proteins: Nck1 (also called Nckalpha) and Nck2, which show partly overlapping functions but also bind distinct targets. The first SH3 domain of Nck2 binds the PxxDY sequence in the CD3e cytoplasmic tail; this binding inhibits phosphorylation by Src kinases, resulting in the downregulation of TCR surface expression. SH3 domains are protein interaction domains that usually bind to proline-rich ligands with moderate affinity and selectivity, preferentially a PxxP motif. They play versatile and diverse roles in the cell including the regulation of enzymes, changing the subcellular localization of signaling pathway components, and mediating the formation of multiprotein complex assemblies. Pssm-ID: 212832 [Multi-domain] Cd Length: 58 Bit Score: 35.49 E-value: 9.20e-03
|
||||||||
| SH3_Alpha_Spectrin | cd11808 | Src homology 3 domain of Alpha Spectrin; Spectrin is a major structural component of the red ... |
12-65 | 9.58e-03 | ||||
Src homology 3 domain of Alpha Spectrin; Spectrin is a major structural component of the red blood cell membrane skeleton and is important in erythropoiesis and membrane biogenesis. It is a flexible, rope-like molecule composed of two subunits, alpha and beta, which consist of many spectrin-type repeats. Alpha and beta spectrin associate to form heterodimers and tetramers; spectrin tetramer formation is critical for red cell shape and deformability. Defects in alpha spectrin have been associated with inherited hemolytic anemias including hereditary spherocytosis (HSp), hereditary elliptocytosis (HE), and hereditary pyropoikilocytosis (HPP). Alpha spectrin contains a middle SH3 domain and a C-terminal EF-hand binding motif in addition to multiple spectrin repeats. SH3 domains are protein interaction domains that bind to proline-rich ligands with moderate affinity and selectivity, preferentially to PxxP motifs. They play versatile and diverse roles in the cell including the regulation of enzymes, changing the subcellular localization of signaling pathway components, and mediating the formation of multiprotein complex assemblies. Pssm-ID: 212742 [Multi-domain] Cd Length: 53 Bit Score: 35.15 E-value: 9.58e-03
|
||||||||
| Blast search parameters | ||||
|